Clover Announces Positive Preliminary Phase I Results for Bivalent RSV Vaccine Candidate SCB-1019 in Initial Young Adult Cohort
-- Bivalent SCB-1019 significantly boosted RSV-A and RSV-B neutralization titers to approximately 6,600 IU/mL (6.4-fold increase) and approximately 46,000 IU/mL (12-fold increase), respectively --
-- Preliminary Phase I data in target older adult population is on track for H2-2024 --
SHANGHAI, April 8, 2024 /PRNewswire/ -- Clover Biopharmaceuticals, Ltd.(Clover; HKEX: 02197), a global commercial-stage biotechnology company committed to unleashing the power of innovative vaccines to save lives and improve health around the world, today announced positive preliminary immunogenicity and safety data in the initial young adult cohort from its Phase I trial evaluating SCB-1019 – the company's bivalent RSV prefusion-stabilized F (PreF)-Trimer subunit vaccine candidate – which is based on Clover's Trimer-Tag vaccine technology platform.
"We are encouraged by the progress and positive preliminary Phase I results for our bivalent RSV PreF vaccine candidate utilizing our validated Trimer-Tag platform, demonstrating broad and significant neutralizing antibody responses against both RSV-A and RSV-B," said Joshua Liang, Chief Executive Officer & Board Director of Clover. "As the first RSV PreF vaccine candidate developed in China to enter the clinical trial stage and now the first to generate clinical data, we look forward to the additional Phase I clinical data in our initial target older adult population in the second half of 2024 as planned."
In the initial cohort enrolling young adults (aged 18-59) in the Phase I trial receiving either SCB-1019 or saline placebo, the preliminary results for geometric mean titers (GMTs) and geometric fold rises (GMFRs) for neutralizing antibodies on Day 0 (pre-vaccination) and Day 28 (post-vaccination) were as follows:
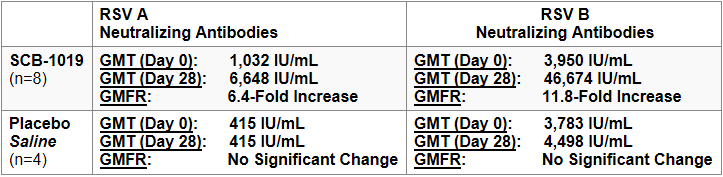
The RSV-A and RSV-B neutralization assays in this study were conducted at a third-party testing laboratory utilizing validated clinical assays and the NIBSC 16/284 reference standard sera, with assay values expressed as international units per milliliter (IU/mL).
Clover's preliminary immunogenicity data across both RSV-A and RSV-B neutralization appear to be in-line or potentially favorable compared to other protein subunit RSV PreF vaccines[1, 2, 3] and are supportive of Clover's bivalent RSV-A/B approach, given that other monovalent RSV-A vaccines have previously observed lower immune responses and/or efficacy against RSV-B [1, 4, 5]. The results also confirm that Clover's PreF antigens in SCB-1019 are in the stabilized prefusion and trimeric form, further supported by exploratory immunogenicity results demonstrating significant increases in Site Ø neutralizing antibody-competitive titers. Additionally, SCB-1019 vaccination did not observe any notable safety or reactogenicity issues in this initial young adult cohort, enabling the planned enrollment of older adults to proceed in the Phase I clinical trial.
The Phase I clinical trial in Australia is a randomized, placebo-controlled study to assess the safety, reactogenicity and immunogenicity of SCB-1019 at multiple dose levels and in different formulations in young and older adults. Additional safety and immunogenicity results in older adults are expected by the second half of 2024.
[1] Icosavax Company Presentations (28-JUN-2022 & 22-MAY-2023) and Press Release (12-DEC-2023)
[2] NIH DS-Cav1 (DOI: 10.1016/S2213-2600(21)00098-9)
[3] Pfizer (DOI: 10.1093/infdis/jiab612)
[4] GSK ACIP Presentation (21-JUN-2023)
[5] Moderna ACIP Presentation (29-FEB-2024)
About Clover
Clover Biopharmaceuticals is a global commercial-stage biotechnology company committed to unleashing the power of innovative vaccines to save lives and improve health around the world. With integrated research and development, manufacturing and commercial capabilities as well as strong partnerships with organizations globally, Clover has a diverse pipeline of candidates that have the potential to meaningfully reduce the burden of vaccine-preventable diseases—and to make more diseases preventable.
Clover Forward-looking Statements
This press release contains certain forward-looking statements and information relating to us and our subsidiaries that are based on the beliefs of our management as well as assumptions made by and information currently available to our management. When used, the words "aim," "anticipate," "believe," "could," "estimate," "expect," "going forward," "intend," "may," "might," "ought to," "plan," "potential," "predict," "project," "seek," "should," "will," "would" and the negative of these words and other similar expressions, as they relate to us or our management, are intended to identify forward-looking statements. Forward-looking statements are based on our current expectations and assumptions regarding our business, the economy and other future conditions. We give no assurance that these expectations and assumptions will prove to have been correct. Because forward-looking statements relate to the future, they are participant to inherent uncertainties, risks and changes in circumstances that are difficult to predict. Our results may differ materially from those contemplated by the forward-looking statements. They are neither statements of historical fact nor guarantees or assurances of future performance. We caution you therefore against placing undue reliance on any of these forward-looking statements. Any forward-looking statement made by us in this document speaks only as of the date on which it is made. Factors or events that could cause our actual results to differ may emerge from time to time.
Clover Biopharmaceuticals
Corporate Communication:Lola Cao
Investor Relations: Michael Tu





